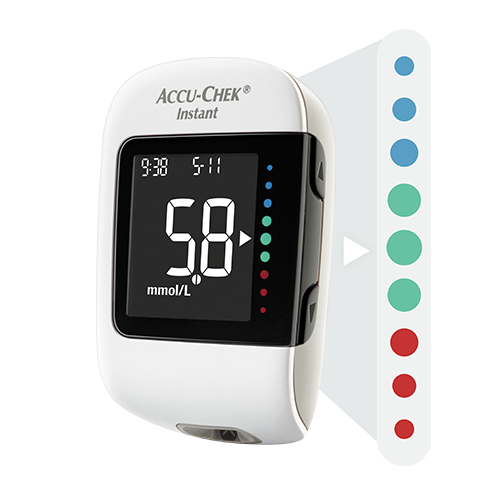
Benefits
- Simpy clear The target range indicator visually helps patients to correctly interpret their blood glucose values1.
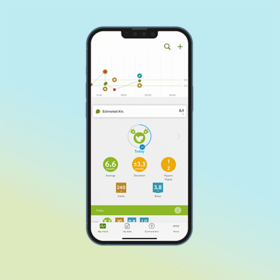
- Simply connected Bluetooth® enabled connectivity allows seamless transfer of blood glucose results to the mySugr® app. This makes it easier for patients to log their diabetes data and share with you ahead of consultations.
- Proven accuracy Fulfills ISO 15197:2013/EN ISO 15197:2015 and delivers even tighter 10/10 accuracy2, for results you can trust.
- Cost effective £5.95 per 50 test strips on the drug tariff*, €8.54 per 50 test strips on Primary Care Reimbursement Service (PCRS)**
Turn numbers into better outcomes<sup>3</sup>
The Accu-Chek Instant meter and mySugr app help your patients better self-manage their diabetes and provide you with the accurate data you need to make personalised treatment decisions for better therapy outcomes.2,3

Resources for you
Connect with a representative
Do you have questions, or are you ready to see a demo? Schedule a call, and we'll show you what we can do for you, your team, and your patients.
Request demoNeed help with your patients' products?
From how-to-videos and FAQs to product manuals, our support resources are here for you and your patients every step of the way.
Product SupportDisclaimers
mySugr app: You must be at least 18 years old to register for the Accu-Chek Account. Please see the manual for intended use.
The Accu-Chek FastClix finger pricker is intended for patient self-monitoring by an individual person only. It must not be used to collect blood in a multipatient setting as it does not incorporate any features to guard against cross infection.
References
*Refers to UK Drug Tariff price as of June 2024. Please refer to www.nhsbsa.nhs.uk for updates.
**Refers to PCRS reimbursable items listed price as of June 2024. For updates please refer to www.sspcrs.ie/druglist/pub
1. Parkin CG et al. Use of an Integrated Tool for Interpretation of Blood Glucose Data Improves Correctness of Glycemic Risk Assessment in Individuals with Type 1 and Type 2 Diabetes. J Diabetes Sci Technol. 2017;11(1):74-82
2. Breitenbeck N, Brown A. Accuracy Assessment of a Blood Glucose Monitoring System for Self-Testing with Three Test Strip Lots Following ISO 15197:2013/EN ISO 15197:2015. J Diabetes Sci Technol. 2017 Jul;11(4):854-855.
3. Debong F, Mayer H, and Kober J. Real-World Assessments of mySugr Mobile Health App. Diabetes Technol Ther. 2019;21(S2):S235– S240.
4. Market research in 5 markets with 151 patients and 153 healthcare professionals. Roche Diabetes Care Whitepaper: The Accu-Chek® Instant meter is easy to use for SMBG from a healthcare professional and patient perspective; Results of a web-based online market research in 5 markets with 151 patients and 153 Healthcare Professionals. 22-July-2021. (Authors: Sigrid-Patricia Roehr, Julia Roetschke, Carola Niklaus).
BGM easier (Accu-Chek Instant Target Range Indicator): Parkin CG, et al. Use of an Integrated Tool for Interpretation of Blood Glucose Data Improves Correctness of Glycemic Risk Assessment in Individuals with Type 1 and Type 2 Diabetes. J Diabetes Sci Technol. 2016 Sep 12.
UK version of mySugr real world evidence: Ide C, et al. Real World Data analysis shows a significant improvement in glycaemic management when using a blood glucose monitor connected with a mobile health application in UK users with Type 2 Diabetes. Diabetes UK Professional Conference Poster Session, Liverpool, 26‑28 Apr 2023, Poster 198
Virtually pain-free: Jendrike N et al. Pain sensation at Fingertips and Palm by using different Blood Glucose Monitoring Systems. Diabetes Technology Meeting. San Francisco, 5-7 November 2009.